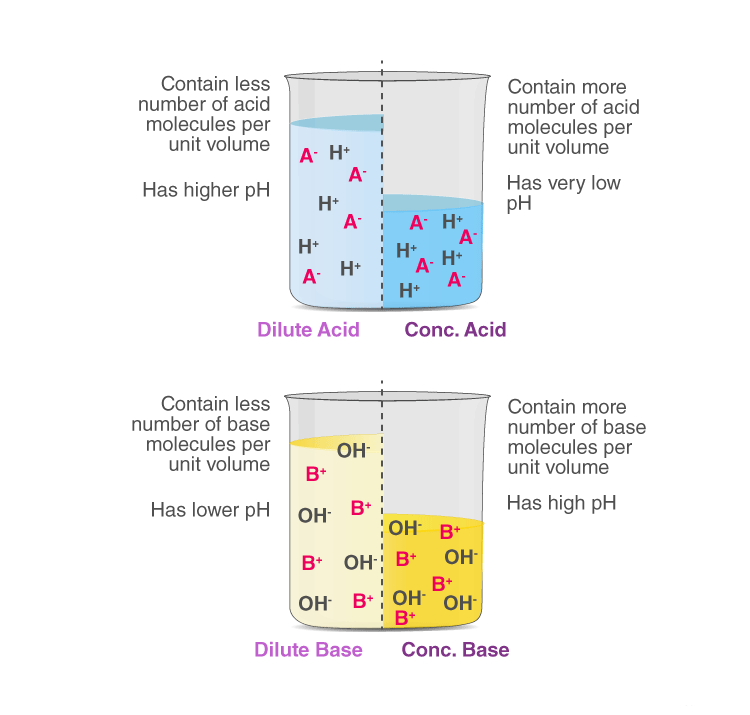
Strong acid or base: When all molecules of a given amount of an acid or a base dissociate completely in water to furnish their respective ions, H+(aq) for acid and OH−(aq) for base).
Weak acid or base: When only a few of the molecules of a given amount of an acid or a base dissociate in water to furnish their respective ions, H+(aq) for acid and OH−(aq) for base).
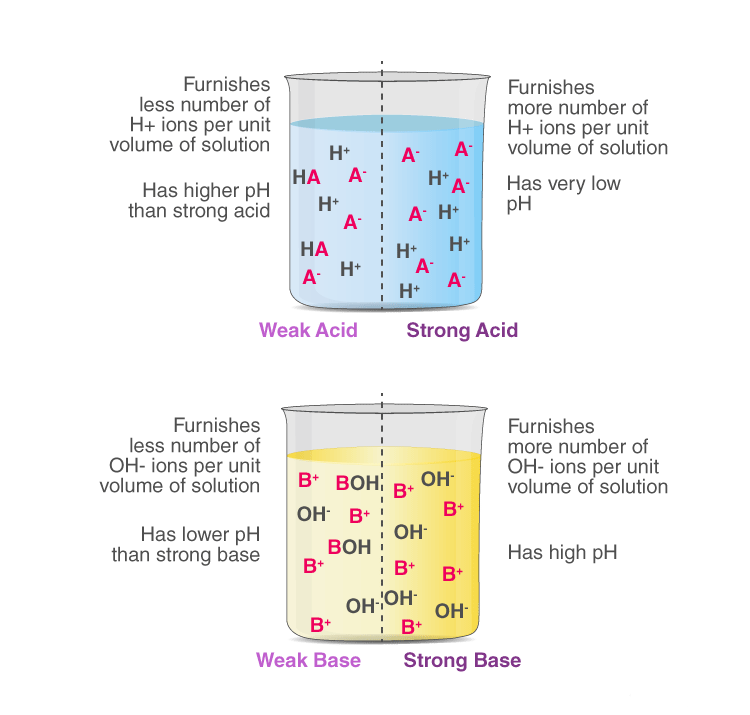
Dilute acid: contains less number of H+(aq) ions per unit volume.
Concentrated acid: contains more number of H+(aq) ions per unit volume.