Properties of electrons, protons and neutrons

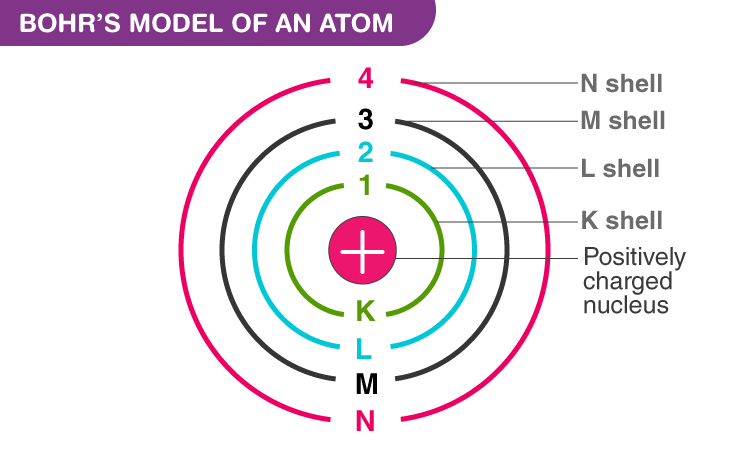
Bohr’s Model of an atom
Bohr came up with these postulates to overcome the objections raised against Rutherford’s model:
- Electrons revolve around the nucleus in stable orbits without emission of radiant energy. Each orbit has a definite energy and is called an energy shell or energy level.
- An orbit or energy level is designated as K, L, M, N shells. When the electron is in the lowest energy level, it is said to be in the ground state.
- An electron emits or absorbs energy when it jumps from one orbit or energy level to another.
- When it jumps from a higher energy level to lower energy level, it emits energy while it absorbs energy when it jumps from lower energy level to higher energy level.