Step-1:
In reverse order, The hydroxide ion deprotonates the aldehyde.

Step-2:
Here Enolate ion 1 adds to the unreacted aldehyde.

Step-3:
Alkoxide ion 2 is protonated by water.
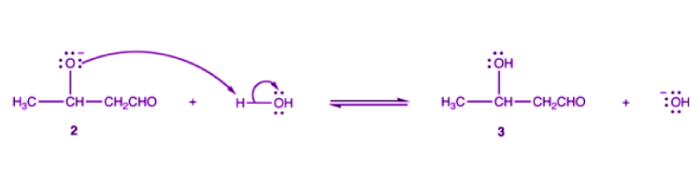
Step-4:
A small amount of aldol is converted into enolate ion (4) by hydroxide ion.

Step-5:
Here Enolate Ion(4) loses a hydroxide ion.

Step 1 to step 3 illustrates the aldol reaction.