Electronic Configuration
Group 1 elements – Alkali metals
| Element | Electronic Configuration |
| Lithium(Li) | 2,1 |
| Sodium(Na) | 2,8,1 |
| Potassium(K) | 2,8,8,1 |
| Rubidium(Rb) | 2,8,18,8,1 |
Group 2 elements – Alkaline earth metals
| Element | Electronic Configuration |
| Beryllium(Be) | 2,2 |
| Magnesium(Mg) | 2,8,2 |
| Calcium(Ca) | 2,8,8,2 |
| Stronium(Sr) | 2,8,18,8,2 |
How Do Metals and Nonmetals React
Metals lose valence electron(s) and form cations.
Non-metals gain those electrons in their valence shell and form anions.
The cation and the anion are attracted to each other by strong electrostatic force, thus forming an ionic bond.
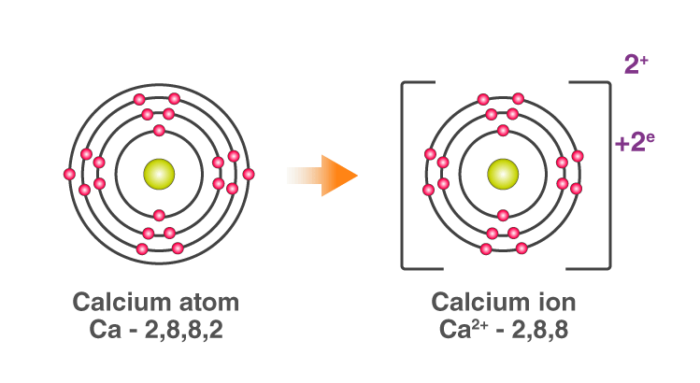
For example: In calcium chloride, the ionic bond is formed by opposite charged calcium and chloride ions.
Calcium atom loses 2 electrons and attains the electronic configuration of the nearest noble gas (Ar). By doing so, it gains a net charge of +2.
The two Chlorine atoms take one electron each, thus gaining a charge of -1 (each) and attain the electronic configuration of the nearest noble gas (Ar).