Base:
- Bases undergo neutralisation reaction with acids.
- They are comprised of metal oxides, metal hydroxides, metal carbonates and metal bicarbonates.
- Most of them are insoluble in water.
Alkali:
- An alkali is an aqueous solution of a base, (mainly metallic hydroxides).
- It dissolves in water and dissociates to give OH− ion.
- All alkalis are bases, but not all bases are alkalis.
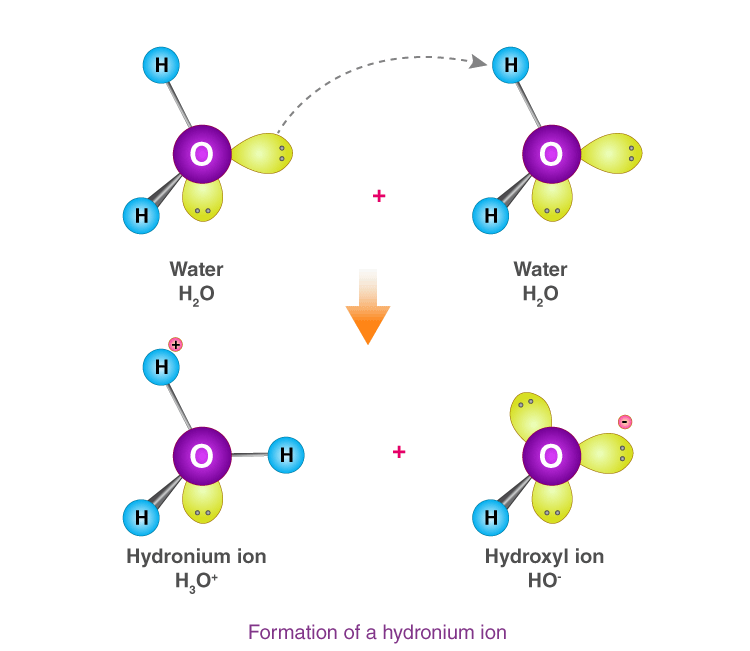
Hydronium ion
Hydronium ion is formed when a hydrogen ion accepts a lone pair of electrons from the oxygen atom of a water molecule, forming a coordinate covalent bond.
Dilution
Dilution is the process of reducing the concentration of a solution by adding more solvent (usually water) to it.
It is a highly exothermic process.
To dilute acid, the acid must be added to water and not the other way round.