Markovnikov’s Rule, also known as Markownikoff’s rule, can be used to describe the outcome of some chemical addition reactions. The Russian chemist Vladimir Vasilyevich Markovnikov first formulated this rule in 1865.
What is Markovnikov’s Rule?
When a protic acid (HX) is added to an asymmetric alkene, the acidic hydrogen attaches itself to the carbon having a greater number of hydrogen substituents whereas the halide group attaches itself to the carbon atom which has a greater number of alkyl substituents.
To simplify the rule, it can also be stated as – “Hydrogen is added to the carbon with the most hydrogens and the halide is added to the carbon with the least hydrogens”.
An example of a reaction that observes Markovnikov’s rule is the addition of hydrobromic acid (HBr) to propene, which is shown below.
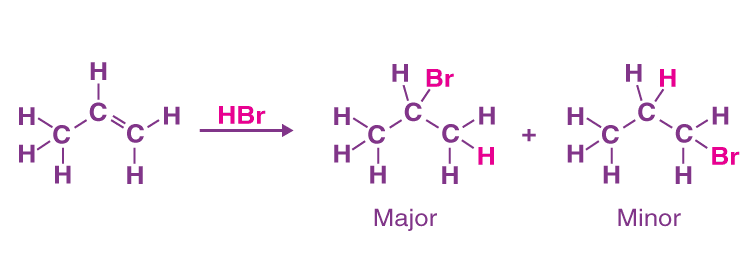
It can be observed from the reaction illustrated above that the majority of the products formed obey Markovnikov’s rule, whereas the minority of the products do not.
Let us consider the addition reaction wherein an alkene reacts with water to give rise to alcohol. This reaction proceeds via the formation of a carbocation. It is observed in this reaction that the hydroxyl group attaches itself to the carbon with more carbon-carbon bonds whereas the hydrogen atom attaches itself to the other carbon in the double bond, which has more carbon-hydrogen bonds.